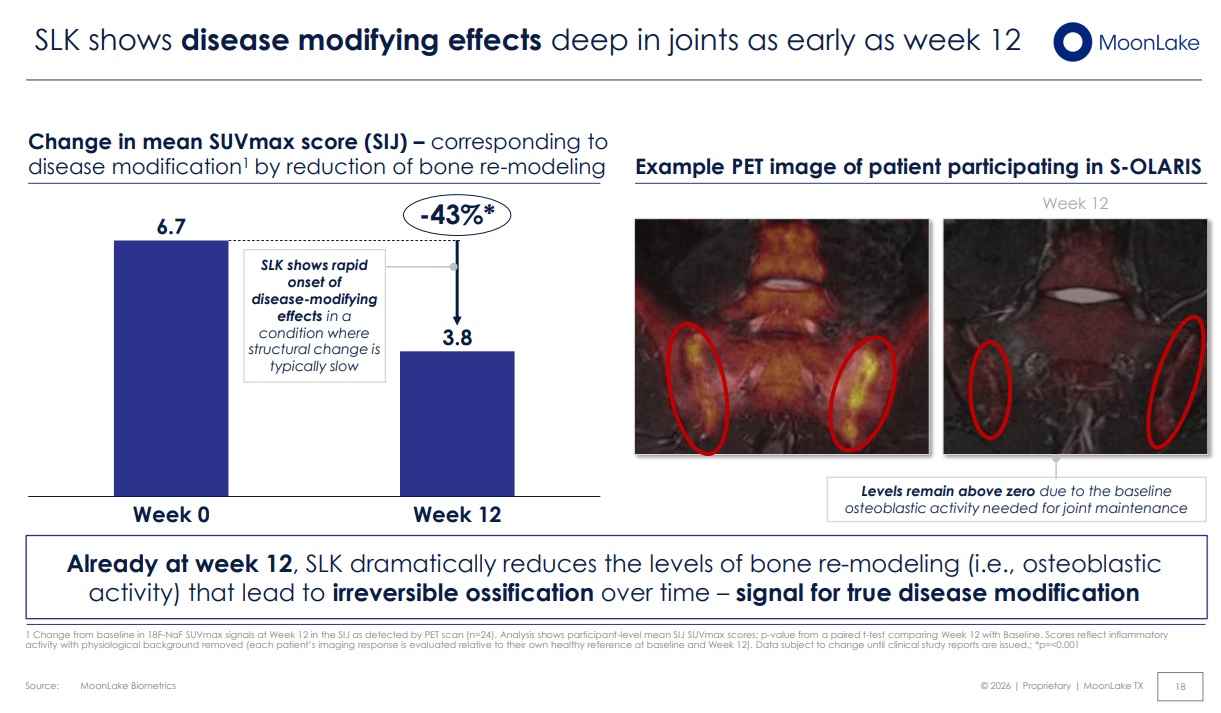
Clinical Trial Imaging in CAR-T Autoimmune Therapies: A Strategic Lever for Recruitment, Response, and Regulatory Confidence
Clinical Trial Imaging in CAR-T Autoimmune Therapies: A Strategic Lever for Recruitment, Response, and Regulatory Confidence
The Next Frontier in Autoimmune Treatment – And Its Biggest Bottleneck
Autoimmune diseases represent one of the largest unmet needs in modern medicine. More than 15–20 million people in the U.S. alone are affected, with many conditions remaining poorly controlled by standard immunosuppressive therapies.
Diseases such as systemic lupus erythematosus (SLE), inflammatory myositis, and rheumatoid arthritis are not only prevalent but increasingly recognized as heterogeneous, biologically complex, and treatment-resistant.
At the same time, the therapeutic landscape is undergoing a paradigm shift.CAR-T cell therapy, once confined to oncology, is now emerging as a potentially curative approach in autoimmune disease, capable of resetting immune tolerance rather than suppressing it.
But despite promising early data, one critical challenge remains: How do you identify the right patients, measure meaningful response, and demonstrate efficacy early enough to de-risk trials?
This is where clinical trial imaging becomes a strategic differentiator, not just a supporting tool.
CAR-T in Autoimmune Disease: From Concept to Clinical Reality
CAR-T therapies in autoimmune indications primarily target autoreactive B cells (e.g., CD19, BCMA), eliminating the root cause of immune dysregulation.
Recent clinical data shows:
- High remission rates (up to 84–95%) across diseases like SLE and myositis
- Complete remission observed in early trials across SLE, systemic sclerosis, and inflammatory myositis
- Rapid immune reset, with normalization of biomarkers and elimination of disease flares
Pipeline momentum is accelerating:
- Over 56 CAR-T trials in autoimmune rheumatic diseases, with the majority actively recruiting
- Non-oncology CAR-T trials growing rapidly, from 2% to 28% of trial starts between 2022–2024
Key indications include:
1. Systemic Lupus Erythematosus (SLE)
- Nearly 1 million patients in the U.S. alone
- High unmet need due to disease heterogeneity and flare unpredictability
2. Inflammatory Myositis
- Rare but severe; often refractory to standard therapy
- CAR-T trials show deep remission and functional recovery
3. Systemic Sclerosis and Related Disorders
- Progressive fibrosis with limited treatment options
- Early CAR-T data suggests immune-driven disease reversal
The Problem: Why CAR-T Autoimmune Trials Are Hard to Execute
Despite clinical promise, autoimmune CAR-T trials face three systemic challenges:
1. Patient Identification and Recruitment
Patient identification and recruitment in autoimmune CAR-T trials remain a significant challenge due to the inherent heterogeneity of these diseases. Variability in disease presentation complicates the development of precise inclusion criteria, while reliance on clinical symptoms alone often lacks the specificity needed to identify appropriate candidates. As a result, many patients undergo multiple lines of unsuccessful therapy before meeting eligibility requirements, delaying enrollment and limiting access to potentially transformative treatments.
2. Measuring Meaningful Efficacy
Measuring meaningful efficacy in autoimmune CAR-T trials is particularly challenging, as traditional endpoints, such as clinical scores and standard biomarkers, are often slow to reflect therapeutic impact. These measures can be subjective and may not accurately capture underlying disease biology, limiting their ability to demonstrate early and mechanistically relevant treatment effects.
3. Demonstrating Early Signal
Demonstrating an early signal of efficacy is increasingly critical in autoimmune CAR-T trials, as investors, sponsors, and regulators expect rapid proof-of-concept supported by quantifiable and reproducible endpoints. Beyond clinical improvement, there is growing emphasis on mechanistic validation, clearly showing how novel therapies are driving biological change at the tissue and disease level.
Without objective, sensitive tools, trials risk delays, underpowered outcomes, and inconclusive results.
The Solution: Imaging as a Strategic Asset in CAR-T Trials
Advanced imaging, particularly MRI-based quantitative analysis, is transforming how autoimmune trials are designed and executed.
Rather than serving as a secondary endpoint, imaging is now enabling:
1. Precision Patient Selection
Imaging can identify subclinical inflammation and tissue involvement that may not be captured clinically.
In myositis, for example:
- MRI has been used to track muscle inflammation and treatment response post CAR-T therapy
- Studies show complete resolution of inflammation on imaging alongside clinical improvement
Impact:
Drives more targeted recruitment by identifying the right patients earlier, reducing screen failure rates. It also enriches study populations with those most likely to respond, improving trial efficiency and outcome quality.
2. Early, Quantitative Readouts of Efficacy
Unlike clinical scores, imaging provides:
- Objective, reproducible biomarkers
- Whole-organ assessment
- Early detection of treatment response
This is critical in CAR-T trials, where:
- Mechanisms are novel (immune reset vs suppression)
- Traditional endpoints may lag behind biological change
Impact:
Enables faster go/no-go decisions by providing early, objective signals of efficacy. It also strengthens interim analyses, ultimately reducing overall trial duration and cost.
3. Mechanistic Validation of Novel MOAs
With emerging approaches such as:
- Dual-target CAR-T (CD19 + BCMA)
- Allogeneic “off-the-shelf” CAR-T
- CAR-T regulatory cells (Tregs)
Sponsors must demonstrate:
- Where and how the therapy is working
- Impact on tissue-level inflammation and damage
Imaging provides a direct window into disease biology, enabling:
- Visualization of immune reset
- Correlation with biomarker and clinical outcomes
Impact:
Supports stronger regulatory narratives by providing clear, mechanism-based evidence of treatment impact. It also enables differentiation in crowded pipelines while increasing confidence among regulators, investors, and other stakeholders.
Why Imaging Matters More in Autoimmune CAR-T Than Oncology
Unlike tumors, autoimmune diseases:
- Are diffuse and systemic
- Lack a single measurable lesion
- Require quantification of inflammation, not just size reduction
This makes imaging not just useful, but essential. In fact, as CAR-T expands into autoimmune indications (currently ~2.75% of trials but rapidly growing), standardized imaging will be critical to scaling development.
IAG: Enabling Precision Imaging in CAR-T Autoimmune Trials
As CAR-T therapies redefine autoimmune treatment, imaging must evolve alongside them. This is where IAG delivers measurable impact. Through advanced imaging analytics platforms such as DYNAMIKA™, IAG enables:
Quantitative Assessment of Inflammation
- Standardized, reproducible MRI biomarkers
- Sensitive detection of early response
Optimized Trial Design
- Imaging-driven inclusion criteria
- Reduced variability across sites
Accelerated Decision-Making
- Early efficacy signals
- Robust interim analyses
Regulatory-Ready Data
- Validated imaging endpoints
- Integration with clinical and biomarker datasets
With deep expertise in inflammatory diseases and partnerships across global pharma, IAG supports trials from protocol design through regulatory submission—ensuring imaging is not an afterthought, but a core strategic asset.
The Future: Imaging as a Competitive Advantage in CAR-T Development
CAR-T is poised to redefine autoimmune care—moving from chronic management to potential cure.
But success will depend on more than biology.
Sponsors who win will be those who can:
- Identify the right patients faster
- Demonstrate efficacy earlier
- Generate robust, regulator-ready evidence
Imaging is central to all three.
About Image Analysis Group (IAG)
Founded in 2007 and headquartered in London, Image Analysis Group (IAG) is the world’s leading specialist Imaging Clinical Research Organization (iCRO). IAG partners with the world’s top pharmaceutical, biotechnology, and clinical research organizations to design, execute, and deliver imaging-based clinical trial endpoints across oncology, inflammatory disease, neurology, musculoskeletal, cardiovascular, and rare disease indications.
IAG’s proprietary DYNAMIKA™ platform is the most advanced cloud-based clinical trial imaging management system available, integrating AI-driven image analysis, centralized quality control, regulatory-compliant reporting, and real-time trial oversight across global multi-center studies. IAG’s global teams – spanning the United Kingdom, United States, European Union, and India — combine deep medical imaging science, therapeutic area expertise, and regulatory intelligence to accelerate drug development timelines and strengthen the evidentiary quality of imaging endpoints in regulatory submissions.
Key Takeaway
As CAR-T pipelines expand across autoimmune indications, integrating advanced imaging is crucial for earlier diagnosis and treatment.
Partner with IAG to de-risk your CAR-T clinical trials and accelerate patient recruitment and readouts. Deliver high-quality, quantitative evidence that strengthens confidence among regulators and stakeholders.
Connect with IAG today to explore how imaging can transform your next autoimmune trial.