How Centralized Imaging Reduces Variability in Immuno-Oncology Trials

The Variability Challenge in Immuno-Oncology Trials
Immuno-oncology (IO) trials have transformed cancer research, introducing therapies that activate the immune system to fight tumors. However, these trials often rely heavily on imaging endpoints, tumor response, progression assessment, and biomarker evaluation, making consistency in imaging analysis critical.
In multi-center studies, variability can arise from differences in imaging equipment, acquisition protocols, reader interpretation, and data processing. These inconsistencies may compromise endpoint reliability, delay regulatory approvals, and introduce bias into trial outcomes. Centralized imaging, supported by core labs, imaging CROs and standardized workflows, has emerged as a strategic solution to reduce variability and strengthen data quality across global studies.
Why Variability Is a Major Concern in IO Trials
Imaging variability is not just a technical challenge, it can fundamentally affect clinical trial conclusions. In oncology studies using RECIST criteria, reader disagreement remains a significant issue. A recent analysis of double-read imaging data across multiple trials found nearly 39% discordance in disease progression assessments, highlighting how subjective interpretation can influence outcomes and survival analyses.
Additional variability stems from differences in imaging protocols and equipment across sites. Research comparing imaging systems across hospitals has shown measurement differences of up to 20–58% in quantitative imaging outcomes when protocols were not standardized.
For IO trials, where subtle tumor changes, pseudoprogression, or delayed responses are common, such inconsistencies can significantly impact endpoint accuracy and decision-making.
The Role of Centralized Imaging in Multi-Center Trials
Centralized imaging refers to the use of standardized acquisition protocols, blinded independent reads, and centralized core lab oversight to ensure consistency across trial sites. By consolidating imaging analysis within a controlled framework, sponsors gain a single source of truth for imaging data.
Centralized approaches reduce inter-reader variability by implementing uniform reading environments and standardized workflows. Expert radiologists trained in trial-specific criteria interpret images consistently, minimizing subjective differences and enhancing confidence in study outcomes.
Beyond interpretation, centralized imaging ensures harmonized acquisition parameters across sites, reducing inconsistencies that arise from different scanners, protocols, or technical expertise.
Key Benefits of Centralized Imaging in IO Trials
1. Improved Endpoint Reliability
Blinded independent central review (BICR) and standardized protocols help ensure accurate measurement of tumor response and progression. Centralized oversight reduces measurement variability and increases consistency across time points and reviewers.
This is particularly important in IO trials, where endpoints such as progression-free survival (PFS) and overall response rate (ORR) rely on precise imaging interpretation.
2. Enhanced Data Quality and Regulatory Confidence
Regulatory agencies increasingly rely on imaging endpoints for drug approvals. Standardized imaging protocols and centralized analysis improve data integrity and ensure consistent evidence across sites, strengthening regulatory submissions.
Centralized imaging also supports compliance with global standards and documentation requirements, reducing the risk of data queries or delays.
3. Reduced Operational Complexity
Managing imaging data across multiple sites can be resource-intensive. Core labs streamline workflows by handling image acquisition guidelines, quality control, data management, and analysis—reducing operational burden on clinical sites.
Centralized systems also enable faster turnaround times and more efficient trial monitoring, supporting agile decision-making.
4. Standardization Across Diverse Trial Environments
Global trials involve diverse equipment, geographic regions, and patient populations. Centralized imaging harmonizes protocols and processing techniques, ensuring comparable results across all sites.
This standardization is essential for multi-modal imaging approaches common in IO trials, including CT, MRI, and PET-based assessments.
5. Advanced Quantitative and AI-Driven Insights
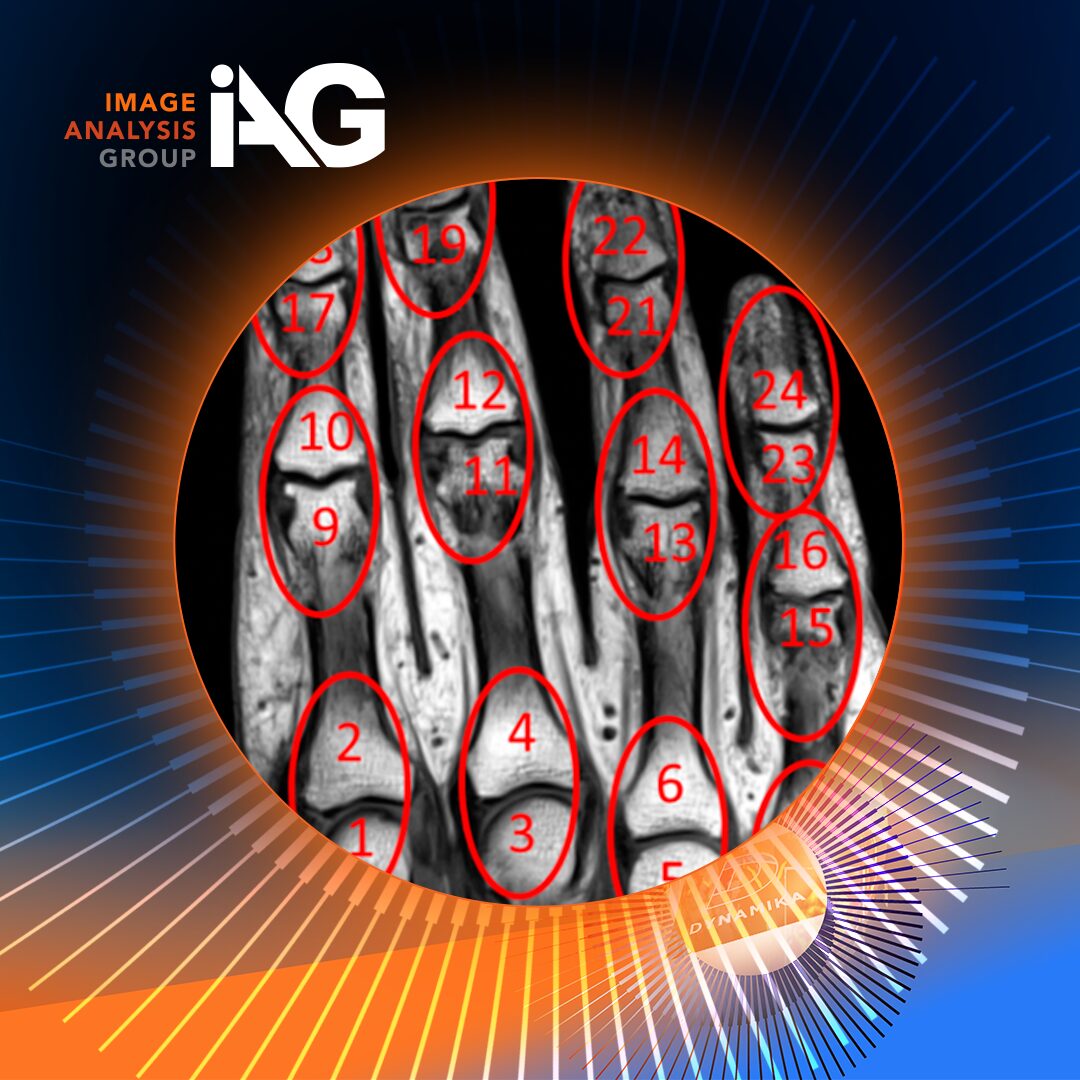
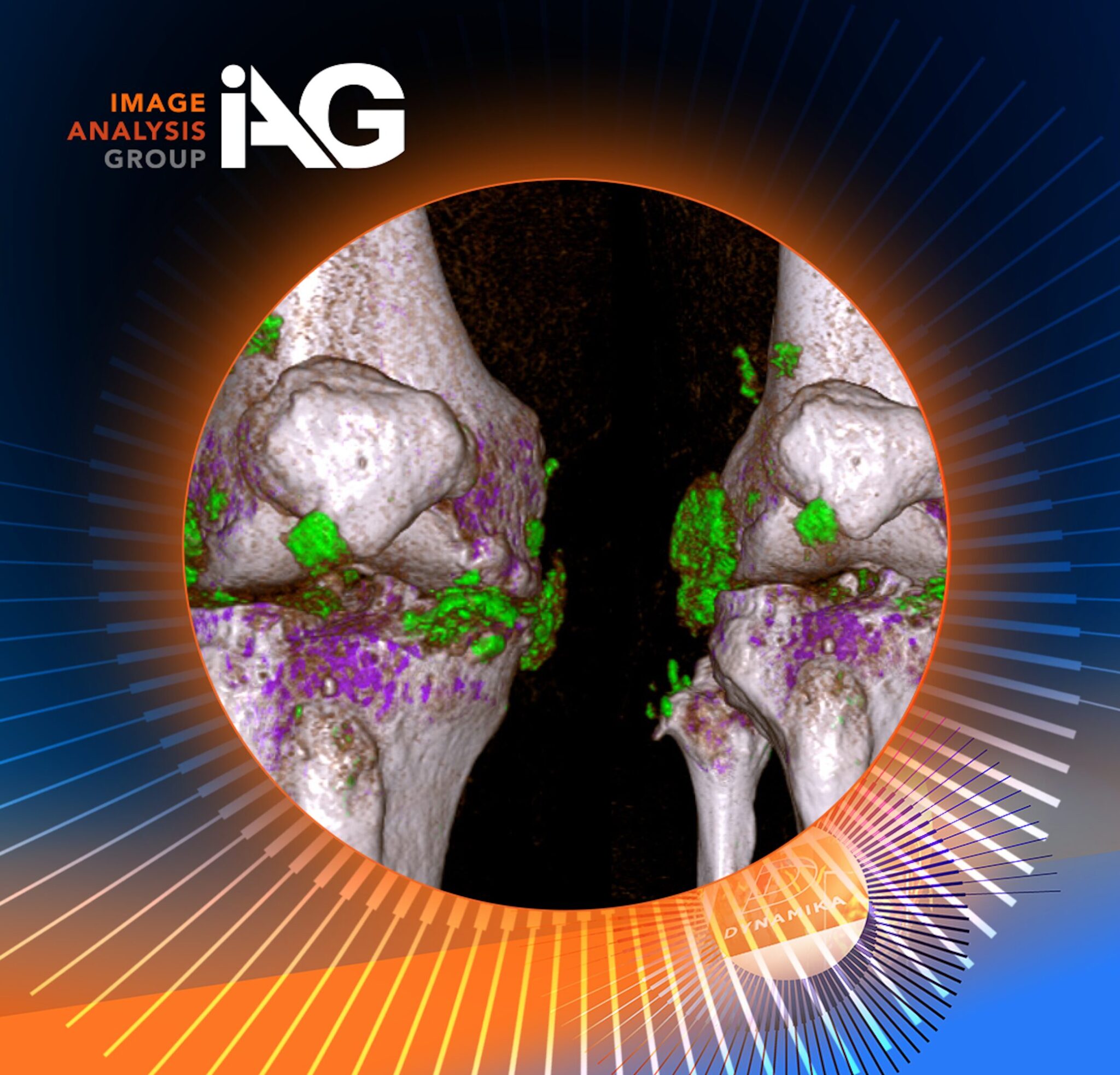
Centralized platforms often integrate advanced analytics and artificial intelligence to enhance image interpretation and biomarker detection. Quantitative imaging enables objective measurement of tumor burden and treatment response, reducing subjective bias and improving reproducibility.
These tools are particularly valuable in IO studies, where nuanced biological responses may not be immediately apparent through conventional analysis.
Best Practices for Implementing Centralized Imaging in IO Trials
Develop Standardized Imaging Protocols Early with AI-Driven Insights
Establishing harmonized acquisition parameters during protocol design minimizes variability from the outset. Clear imaging guidelines ensure consistent data capture across sites.
Utilize Blinded Independent Central Review
Parallel reads with adjudication help reduce bias and improve reliability in progression assessments. Standardized reading environments further support consistency.
Integrate Advanced Imaging Technologies
Leveraging quantitative imaging and AI tools can enhance sensitivity and reduce observer variability, particularly for complex IO endpoints.
Prioritize Training and Site Support
Providing detailed training for site staff ensures adherence to imaging protocols and reduces variability related to human factors.
The Strategic Value of Centralized Imaging for Sponsors
For biotech and pharma sponsors, centralized imaging is not simply a technical enhancement; it is a strategic investment in trial success. High-quality imaging data supports more accurate efficacy assessments, faster decision-making, and improved confidence among regulators and stakeholders.
As IO trials become more complex and globally distributed, centralized imaging helps sponsors manage operational risk while maintaining scientific rigor. By reducing variability, centralized workflows enhance reproducibility, improve trial efficiency, and ultimately accelerate the path to market for innovative therapies.
A Critical Component of Modern IO Trials
Multi-center immuno-oncology trials demand precision, consistency, and scalability. Imaging variability remains one of the most significant challenges affecting endpoint reliability and regulatory outcomes.
Centralized imaging. through standardized protocols, expert analysis, and advanced analytics, offers a proven strategy to reduce variability and strengthen trial integrity. For sponsors seeking to deliver high-quality evidence in increasingly complex global studies, centralized imaging is no longer optional; it is essential.
Ready to Reduce Imaging Variability in Your Trials?
Discover how advanced centralized imaging solutions can optimize your IO trial workflows, improve endpoint reliability, and accelerate study timelines.
Book a personalized DYNAMIKA™ demo today to see how cutting-edge imaging analytics can enhance your clinical trial strategy and drive more confident decision-making.
About Image Analysis Group (IAG)
Image Analysis Group (IAG) is a global imaging clinical research organization (iCRO) focused on de-risking and accelerating drug development through advanced imaging science and AI. IAG designs and runs imaging-centric clinical trials across oncology, immunology, neurology, rare diseases, musculoskeletal conditions, fertility and women’s health, partnering closely with biotech and pharma sponsors from early proof-of-concept through pivotal and real-world evidence studies.
Through its proprietary, cloud-native DYNAMIKA™ platform, IAG integrates centralized imaging workflows, blinded independent review, expert readers and AI-powered imaging biomarkers to deliver high-quality, regulator-ready imaging endpoints and decision-enabling insights that optimize trial design, reduce operational risk and unlock the full value of imaging data.