Quantitative Imaging in Arthritis Clinical Trials: Redefining How Inflammation Is Measured

Quantitative Imaging in Arthritis Clinical Trials: Redefining How Inflammation Is Measured
Arthritis and inflammatory joint diseases remain among the most complex therapeutic areas in clinical development. Characterized by heterogeneous pathophysiology, fluctuating disease activity, and variable treatment response, these conditions present significant challenges for clinical trial design and endpoint selection. Traditional measures, such as clinical scoring systems, patient-reported outcomes, and systemic biomarkers, often fail to capture the full picture of underlying disease activity.
Increasingly, advanced imaging, particularly quantitative MRI, is emerging as a critical tool to bridge this gap. By enabling precise, non-invasive assessment of inflammation at the tissue level, imaging is transforming how clinical trials in arthritis are designed, executed, and interpreted.
IAG is at the forefront of this evolution, whose proprietary imaging technologies and scientific leadership are redefining how inflammation is measured in clinical research.
Imaging as a Biomarker in Arthritis Clinical Trials
In arthritis trials, accurately assessing inflammation is essential, not only for patient stratification but also for evaluating therapeutic efficacy. Synovitis, or inflammation of the synovial membrane, is a key pathological feature across multiple forms of arthritis and a primary driver of joint damage.
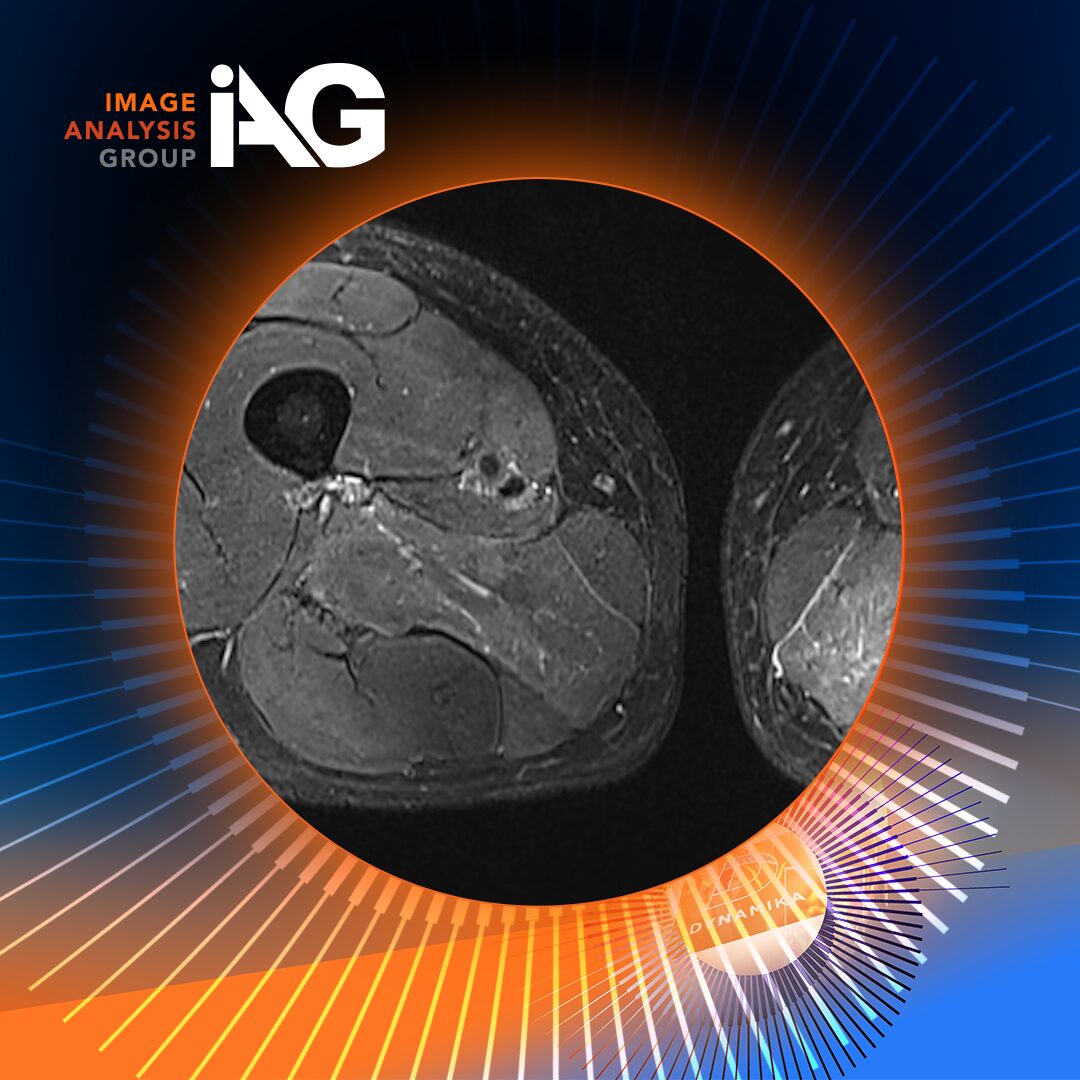
Conventional imaging approaches, including static MRI, provide structural insights but lack the sensitivity to capture dynamic inflammatory processes. This is where Dynamic Contrast-Enhanced MRI (DCE-MRI) has become particularly valuable.
DCE-MRI tracks the uptake and washout of contrast agents in tissues over time, generating time-intensity curves that reflect vascularity and capillary permeability, both hallmarks of active inflammation. However, the real innovation lies not just in acquiring these images, but in quantifying them.
Quantitative Imaging and DEMRIQ: A New Standard for Inflammation Assessment
To move beyond qualitative interpretation, advanced computational methods are required. A study published in the American College of Rheumatology, co-authored by IAG’s CEO, Dr. Olga Kubassova, demonstrates the power of DCE-MRI Quantification (DEMRIQ) in assessing synovial inflammation across multiple arthritis subtypes.
DEMRIQ leverages automated, voxel-based analysis to extract quantitative parameters such as:
- Initial Rate of Enhancement (IRE): Reflecting early inflammatory activity
- Maximum Enhancement (ME): Indicating the degree of vascular permeability
- Enhancing Volume (Nvoxel): Representing the extent of inflamed tissue
Composite metrics such as IRExNvoxel and MExNvoxel further integrate these dimensions, providing a comprehensive measure of both the intensity and volume of inflammation.
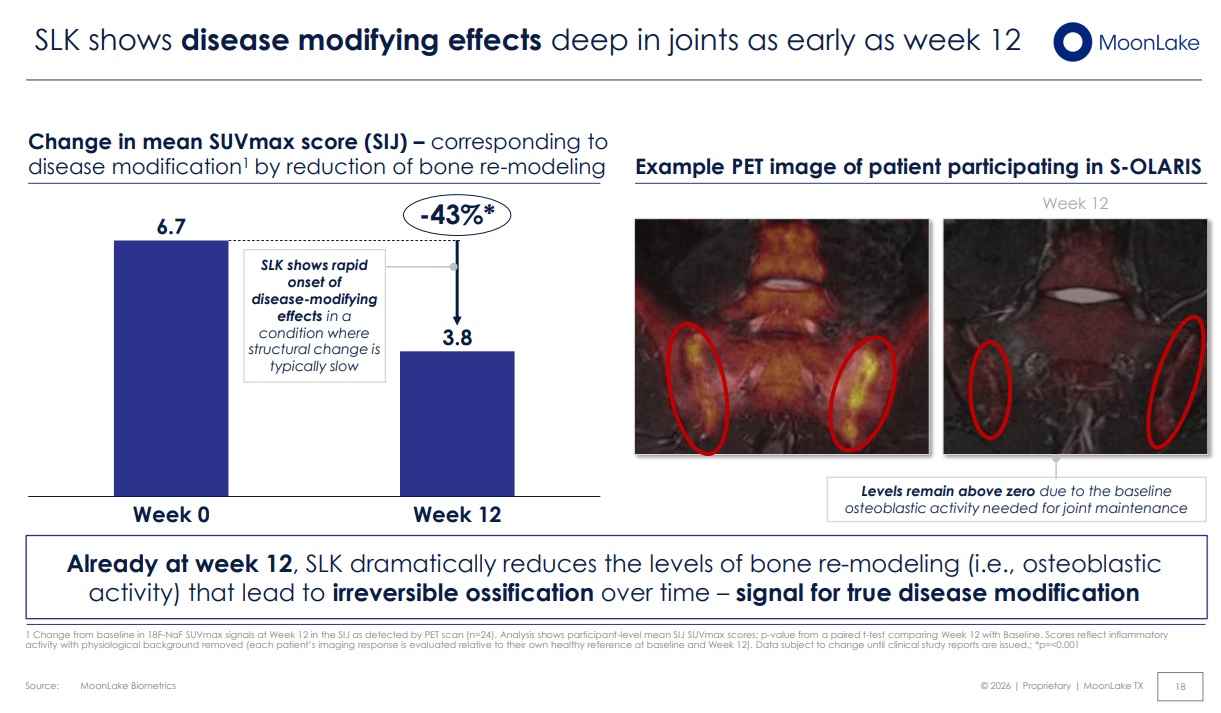
Crucially, the study found strong correlations between DEMRIQ parameters and established inflammatory biomarkers, including synovial fluid interleukin-6 (IL-6), C-reactive protein (CRP), and immune cell counts.
This establishes quantitative imaging not just as a supportive tool, but as a validated surrogate marker of inflammation, capable of reflecting both local and systemic disease activity.
Early Detection and Longitudinal Monitoring of Inflammation
One of the most significant advantages of quantitative imaging is its ability to detect inflammation early, often before structural damage becomes apparent.
In clinical trials, this has several implications:
- Earlier patient stratification: Identifying patients with active inflammation who are more likely to respond to targeted therapies
- Sensitive endpoints: Detecting subtle changes in inflammation over shorter timeframes
- Reduced trial duration and cost: By enabling earlier readouts of therapeutic efficacy
The study highlights that specific DEMRIQ parameters, particularly IRExNvoxel, are highly sensitive to inflammatory cell infiltration and may serve as early indicators of disease activity.
This positions quantitative imaging as a powerful tool for adaptive trial designs, where early signals can inform dose adjustments, cohort expansion, or go/no-go decisions.
Application Across Key Arthritis Indications
Clinical trials in arthritis encompass a range of disease subtypes, each with distinct inflammatory profiles. The study provides valuable insights into how imaging biomarkers perform across three major categories:
1. Rheumatoid Arthritis (RA)
Rheumatoid arthritis is a systemic autoimmune disease characterized by persistent synovitis and joint destruction. The study demonstrated strong correlations between DEMRIQ parameters and both synovial and systemic inflammatory markers in RA patients.
Notably, associations with IL-6, a key cytokine in RA pathogenesis, highlight the potential of imaging to reflect underlying biological pathways and support mechanism-of-action studies.
2. Psoriatic and Seronegative Arthritis
In seronegative arthritis, including psoriatic arthritis, inflammation patterns differ from RA. The study observed higher volumetric inflammation metrics (e.g., MExNvoxel), suggesting that imaging can capture disease-specific inflammatory signatures.
This has important implications for differentiating patient populations and tailoring therapeutic strategies in clinical trials.
3. Osteoarthritis (OA)
Traditionally considered a degenerative disease, osteoarthritis is now recognized to have an inflammatory component. While correlations between imaging and systemic biomarkers were less pronounced in OA, quantitative MRI still demonstrated associations with structural and synovial changes.
This reinforces the role of imaging in understanding disease progression and identifying subpopulations with inflammatory phenotypes.
From Imaging Data to Clinical Insight: The IAG Advantage
While the value of DCE-MRI is well established, its implementation in clinical trials has historically been limited by complexity, variability, and analysis burden.
This is where IAG differentiates itself.
Through its proprietary DYNAMIKA™ platform, IAG delivers fully automated, standardized analysis of DCE-MRI data, transforming raw imaging into reproducible, regulatory-grade biomarkers. The software performs voxel-by-voxel analysis, applies motion correction, and generates color-coded enhancement maps, enabling precise quantification of synovial inflammation.
Key advantages include:
- High reproducibility: With reported inter- and intra-reader reliability exceeding 0.
- Operational efficiency: Reducing analysis time and minimizing user dependency
- Scalability: Supporting multi-center, global clinical trials
- Regulatory alignment: Providing objective, quantifiable endpoints suitable for submission
These capabilities have made IAG a trusted imaging partner for leading pharmaceutical companies, including Lilly, Roche, Novo Nordisk, and Amgen.
Enabling the Next Generation of Inflammation-Driven Trials
As the industry shifts toward precision medicine, the demand for sensitive, objective biomarkers continues to grow. Imaging. particularly when combined with advanced analytics, offers a unique opportunity to directly visualize and quantify disease processes in vivo.
Quantitative MRI biomarkers like DEMRIQ are not only enhancing our understanding of arthritis pathophysiology but also enabling more efficient and informative clinical trials. By integrating imaging with clinical and molecular data, sponsors can gain a multidimensional view of treatment response, ultimately accelerating drug development and improving patient outcomes.
Key Takeaway
Imaging is no longer a secondary endpoint in arthritis clinical trials, it is becoming central to how inflammation is measured, monitored, and understood.
With proven correlations to biological markers, sensitivity to early disease changes, and applicability across multiple arthritis subtypes, quantitative DCE-MRI is redefining the standard for inflammation assessment.
Backed by scientific leadership, peer-reviewed validation, and cutting-edge technology, IAG stands at the forefront of this transformation.
To explore how advanced imaging and quantitative biomarkers can de-risk your arthritis clinical trials and deliver deeper insights into treatment response, connect with IAG’s scientific team or request a demo of DYNAMIKA™ today.