Psoriatic Arthritis Imaging Advancement: A New Scoring System & Global Collaboration
Psoriatic Arthritis Imaging Advancement: A New Scoring System & Global Collaboration
Psoriatic arthritis imaging (PsA) is a complex, heterogeneous inflammatory disease, one that has long challenged both clinicians and drug developers seeking objective, reproducible measures of disease activity.
This challenge is now being addressed through a new generation of advanced imaging-based scoring systems, driven by global collaboration between industry, academic leaders, and specialized imaging clinical research organizations (CROs).
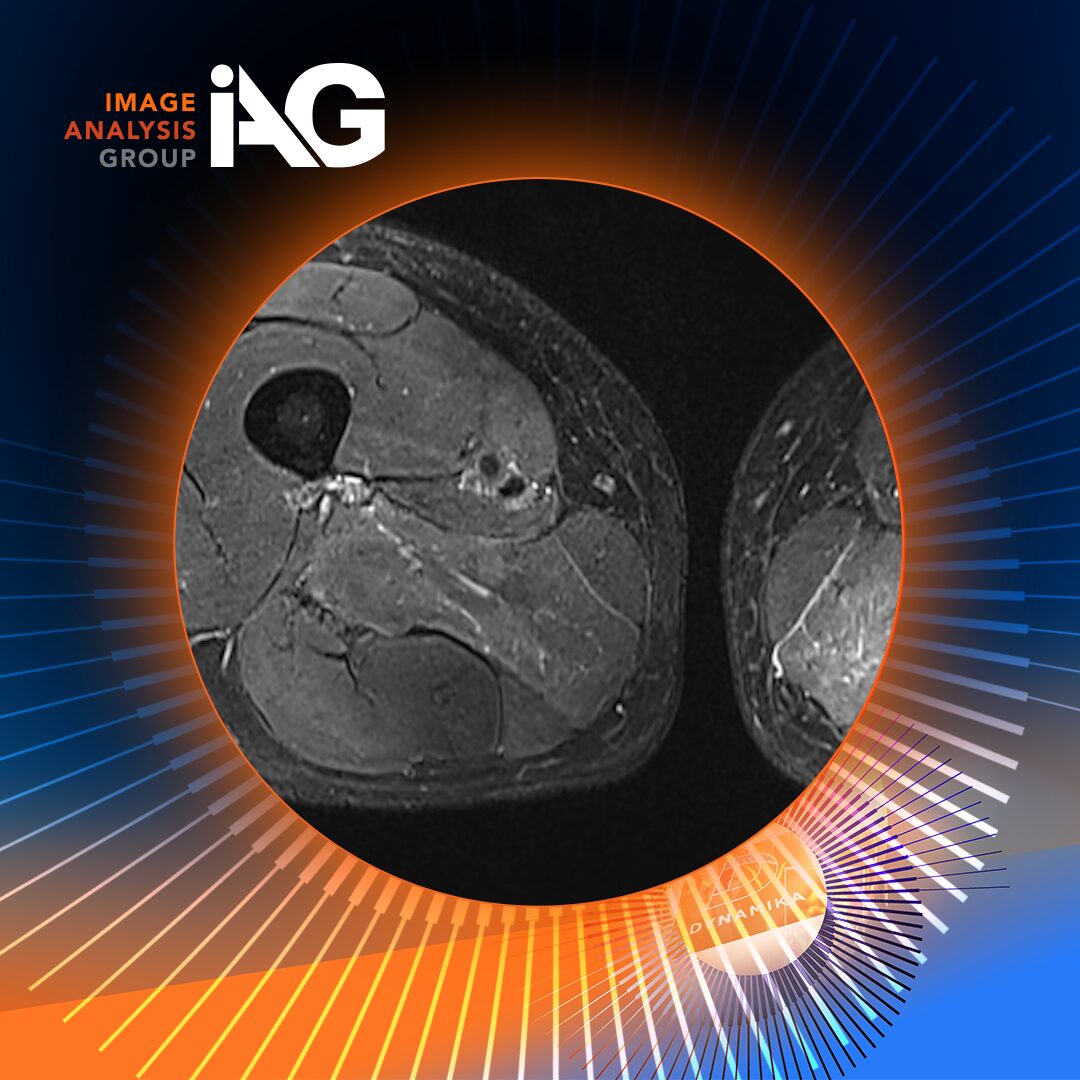
Recent results from the Phase 4 MOSAIC study mark a pivotal moment in PsA research, demonstrating how whole-body MRI (WB-MRI)–based scoring can more comprehensively capture both peripheral and axial inflammation. At the center of this advancement is Image Analysis Group (IAG), whose scientific leadership and imaging expertise continue to redefine how inflammatory disease is measured in clinical trials.
Moving Beyond Traditional PsA Assessment
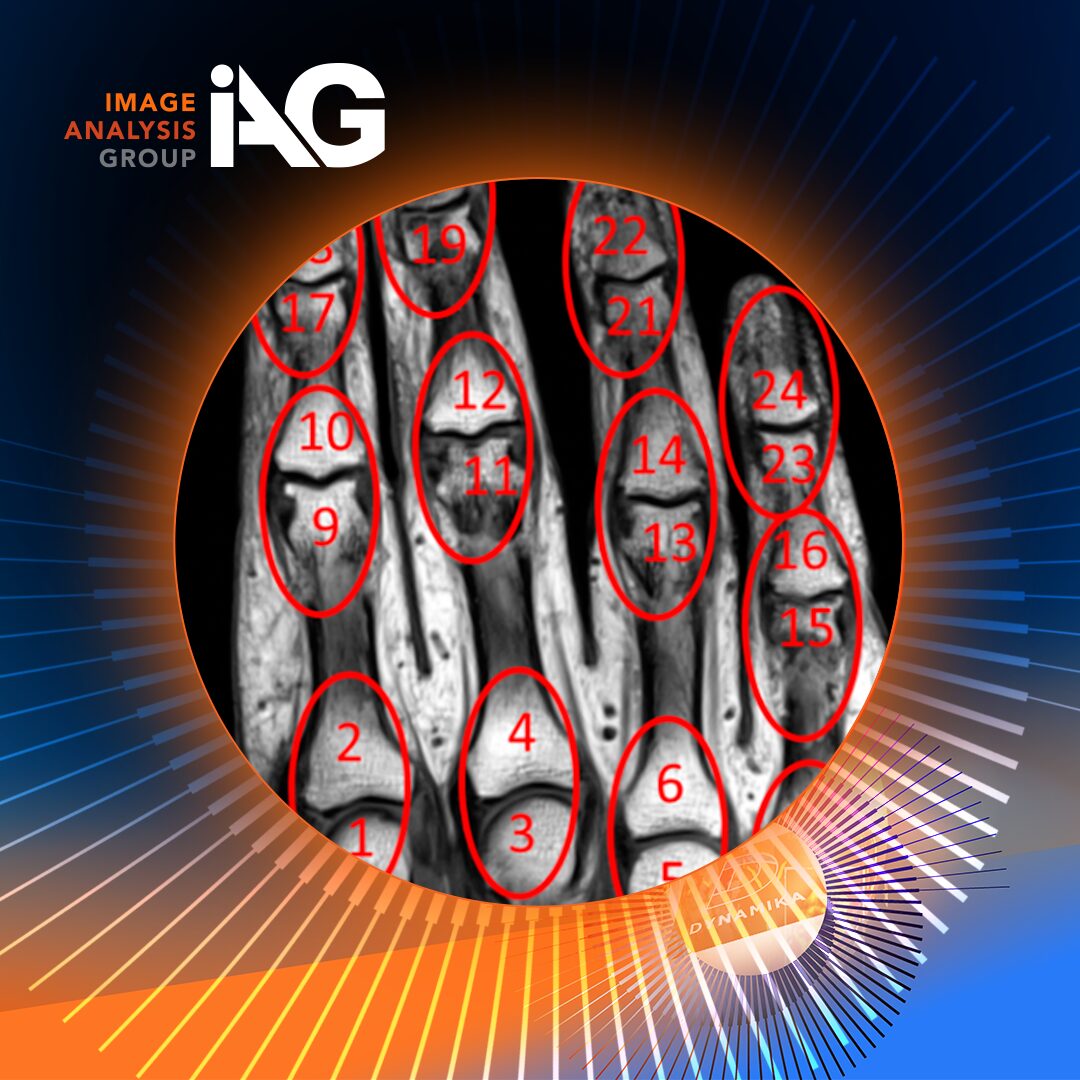
PsA clinical trials rely heavily on clinical indices and localized imaging tools, such as PsAMRIS, which focus primarily on the hands. While valuable, this approach can underestimate disease burden, particularly in PsA, where inflammation often extends across joints, entheses, and the axial skeleton.
The introduction and application of whole-body MRI scoring systems, including OMERACT MRI-WIPE and the Canada-Denmark (CANDEN) Spine Scoring System, represent a major step forward. These tools enable objective, standardized assessment of inflammation across the entire musculoskeletal system, capturing disease manifestations that are otherwise difficult to evaluate through physical exam alone.
In the MOSAIC study, WB-MRI revealed statistically significant reductions in total peripheral and axial inflammation following treatment, offering one of the most comprehensive imaging-based evaluations of PsA response to therapy to date. Notably, CANDEN proved especially sensitive in detecting spinal inflammation, an area often underrepresented by traditional scoring methods.
Scientific Leadership Through Collaboration
The success of this work reflects an exceptional global collaboration between IAG team, sponsor Amgen, and internationally recognized key opinion leaders (KOLs) in rheumatology and musculoskeletal imaging. The study brought together experts from leading institutions across Europe and North America, including pioneers in MRI methodology and PsA disease assessment.
IAG played a central role in this collaboration, providing blinded, standardized image analysis; implementing validated scoring systems; and ensuring methodological rigor across multiple international sites. As both a scientific contributor and imaging CRO partner, IAG helped bridge the gap between imaging innovation and clinical trial execution.
This partnership underscores a broader industry shift: pharmaceutical sponsors increasingly recognize that advanced imaging endpoints require not just technology, but deep disease-area expertise and operational excellence. IAG’s long-standing relationships with imaging KOLs and its active involvement in OMERACT-driven scoring development uniquely position the organization to meet this need.
Why This Matters for Drug Development
For sponsors developing therapies in PsA and related inflammatory diseases, these advances carry significant implications. More sensitive and anatomically comprehensive imaging endpoints can:
- Detect treatment effects earlier in development
- Differentiate efficacy across disease domains
- Support mechanism-of-action insights
- Strengthen regulatory and publication strategies
In MOSAIC, WB-MRI outcomes complemented clinical measures and revealed meaningful differences based on baseline disease activity, demonstrating the potential of imaging biomarkers to guide patient stratification and trial design.
As regulatory agencies and payers increasingly emphasize objective, data-driven evidence, robust imaging endpoints are becoming essential rather than optional. CROs with specialized imaging capabilities and scientific credibility will play a critical role in enabling this shift.
Forefront of Imaging Clinical Research Innovation
With decades of experience in musculoskeletal imaging, inflammatory disease trials, and global multi-reader studies, Image Analysis Group continues to set the standard for imaging CRO performance. The MOSAIC study reinforces IAG’s role not only as a service provider, but as a strategic scientific partner, one capable of advancing novel endpoints while maintaining the highest standards of quality and compliance.
Through its collaboration with Amgen and the world’s leading PsA experts, IAG has helped validate imaging approaches that are likely to influence future clinical trials and, ultimately, clinical practice. As whole-body MRI and advanced scoring systems gain wider adoption, IAG remains at the forefront, translating imaging innovation into actionable clinical trial insights.
The future of PsA research is clearer, more comprehensive, and more objective—and imaging-led innovation is driving the way forward.
About Image Analysis Group (IAG)
Image Analysis Group (IAG) is a global imaging clinical research organization (iCRO) focused on de-risking and accelerating drug development through advanced imaging science and AI. IAG designs and runs imaging-centric clinical trials across oncology, immunology, neurology, rare diseases, musculoskeletal conditions, fertility and women’s health, partnering closely with biotech and pharma sponsors from early proof-of-concept through pivotal and real-world evidence studies.
Through its proprietary, cloud-native DYNAMIKA™ platform, IAG integrates centralized imaging workflows, blinded independent review, expert readers and AI-powered imaging biomarkers to deliver high-quality, regulator-ready imaging endpoints and decision-enabling insights that optimize trial design, reduce operational risk and unlock the full value of imaging data.