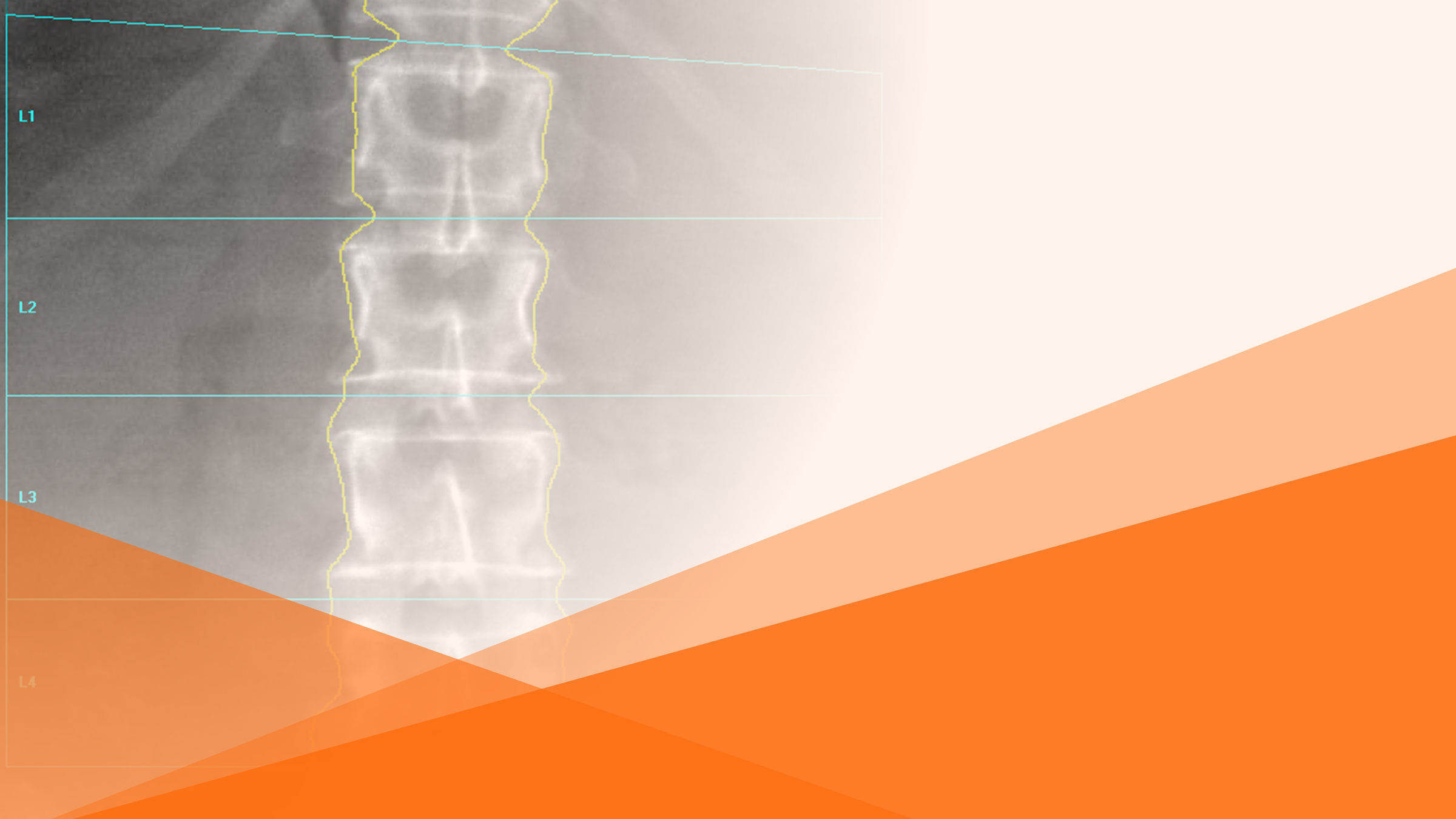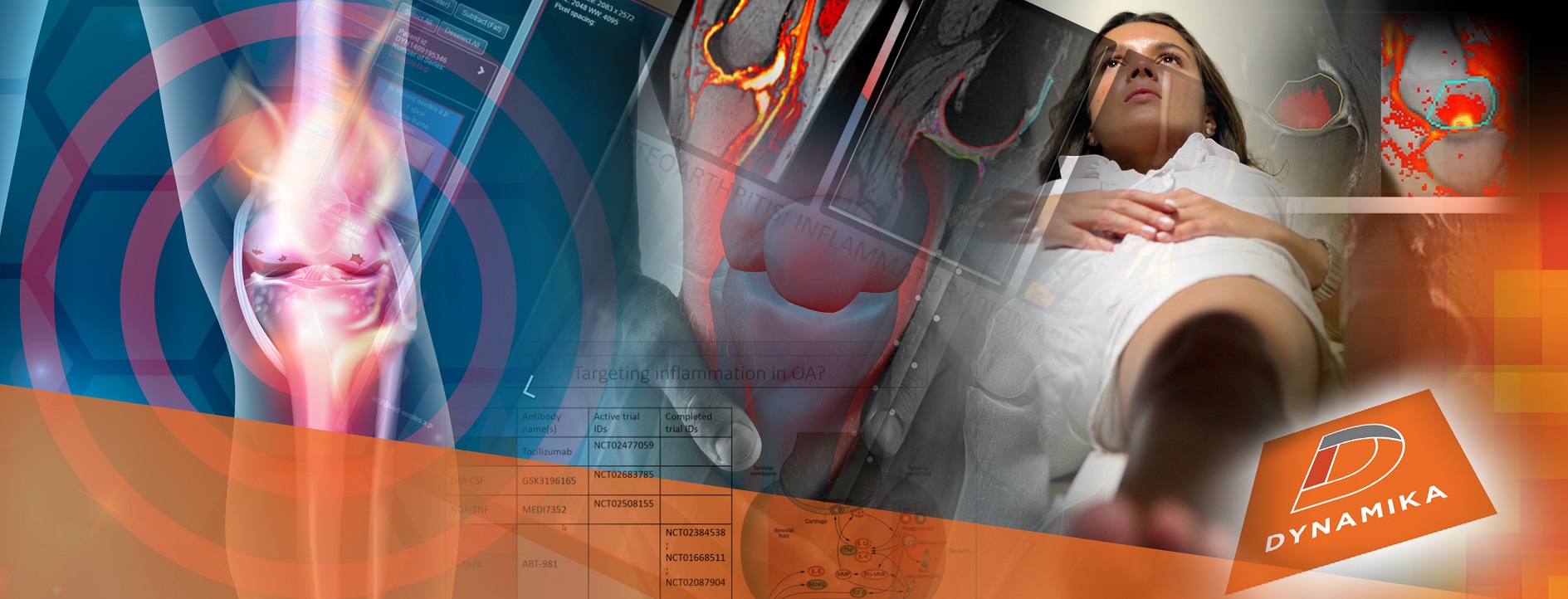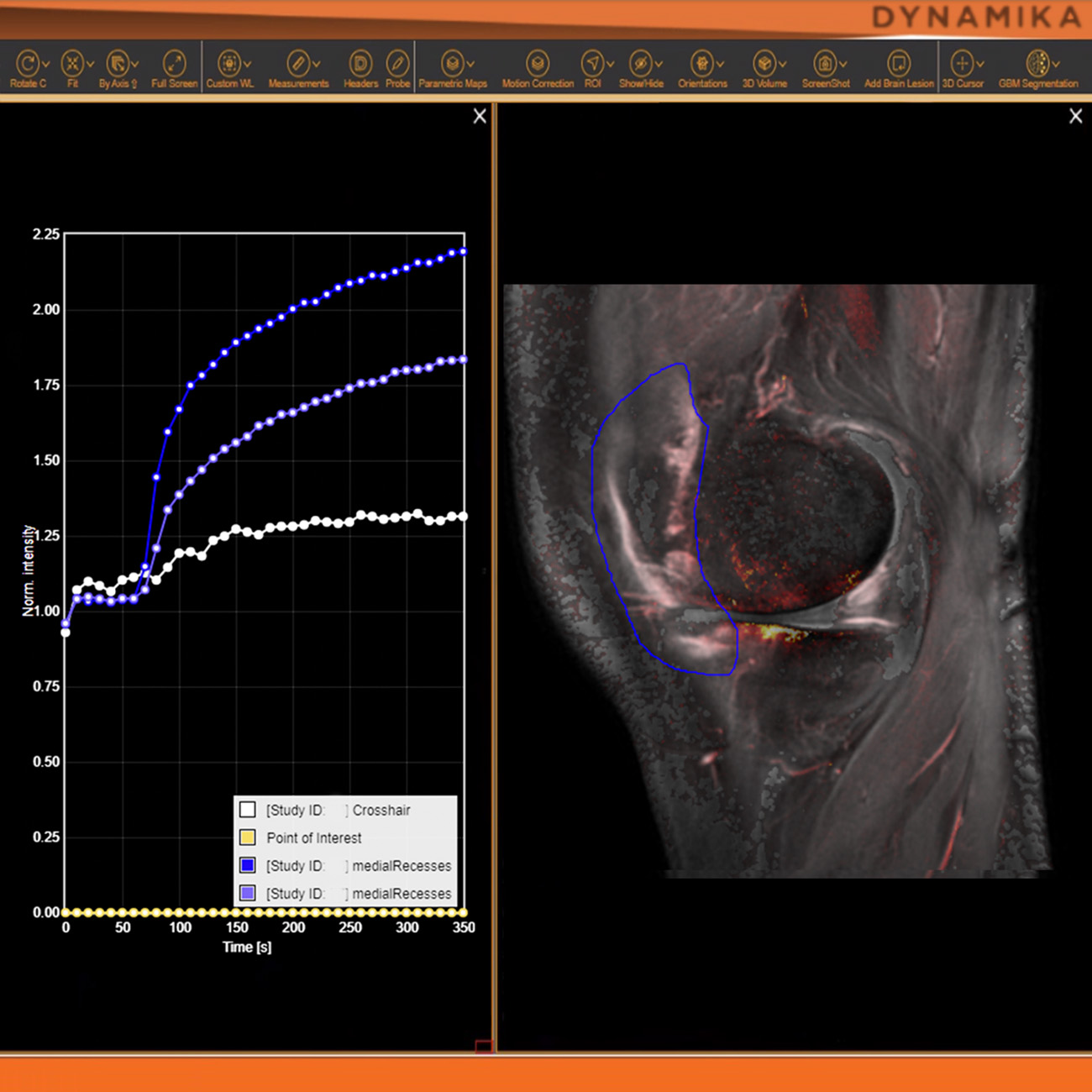

Join Prof. Boesen, IAG’s Sn Theraputic Area Lead and Collaborators to Discuss the Role of AI in Imaging
FOR IMMEDIATE RELEASE
Renowned A.I. expert, Prof. Mikael Boesen, MD, PhD will address OARSI community and drug development companies in upcoming webinar on the Role of Artificial Intelligence in Imaging.
London, 08 May 2024 – As the pharmaceutical industry continues to leverage cutting-edge technologies to enhance drug development processes, the integration of artificial intelligence (AI) in medical imaging emerges as a pivotal advancement.
In an effort to provide crucial insights tailored to Chief Medical Officers (CMOs) in drug development companies, Prof. Mikael Boesen, a distinguished expert in AI will deliver a talk during upcoming webinar titled
“Role of Artificial Intelligence in Imaging: Implications for Drug Development.”
Scheduled for Wednesday, May 15, 2024, from 8:00 to 9:00 am ET, this webinar will serve as a platform for scientist and industry leaders to explore the transformative potential of AI in medical imaging within the context of drug development.
Mikael Boesen will offer strategic perspectives and actionable insights to empower dru development experts in navigating this evolving landscape.
About Prof. Mikael Boesen
With extensive experience in AI research and its intersection with healthcare, Prof. Mikael Boesen brings a wealth of knowledge to the discussion. Currently Professor of MSK Radiology & AI / Co-Founder RAIT.dk and CLIC-project.org, Dr. Boesen has played a pivotal role in advancing AI-driven solutions for medical imaging, with a focus on enhancing diagnostic accuracy, accelerating clinical trials, and optimizing treatment strategies.
During the webinar, participants can expect to gain insights into:
- The role of AI in streamlining imaging processes in drug development
- Leveraging AI-powered image analysis for enhanced efficacy and safety assessments in clinical trials
- Strategies for integrating AI technologies into existing workflows to drive efficiency and innovation
- Regulatory considerations and best practices for deploying AI solutions in drug development settings
The webinar will feature other speakers and collaborators, an interactive Q&A session, providing attendees with the opportunity to engage directly with the speakers and delve deeper into key topics and challenges.
We invite Chief Medical Officers, medical directors, and other senior executives involved in drug development and clinical research within pharmaceutical companies to follow-up with prof Boesen by emailing your questions to imaging.experts@ia-grp.com.
Registration for the webinar is now open: https://oarsi.org/education/oarsi-live-webinar-series
About Image Analysis Group (IAG)
Learn more: ww.ia-grp.com
Reach out: imaging.experts@ia-grp.com
Follow the Company: Linkedin