Image Analysis Group (IAG) is a leading Imaging Clinical Research Organization (iCRO) and valued partner to life sciences companies.
Our mission is to improve patient outcomes by delivering better, faster clinical trials to pharma and biotech utilizing our Digital Data Management infrastructure DYNAMIKA, global operational team and therapeutic expertise.
Our team, well-connected network of sites and radiology readers have delivered over 700 clinical trials, since 2007.
Our vision is to be the partner of choice for pharma and biotech in developing better medicines, faster.
IAG’s foundation is in medical software development, trial endpoints, use of exploratory biomarkers and Artificial Intelligence applied to assess drug efficacy and safety.
We built our own platform for data management and central review – DYNAMIKA. It is the industry leading solution, if you need to maximize efficiencies of handling imaging data in multi-centre clinical trials, fast site set-up and monitoring, image review.
DYNAMIKA is the modern compliant and secure platform, essential to any trial that will use imaging for patient selection or drug efficacy assessment.
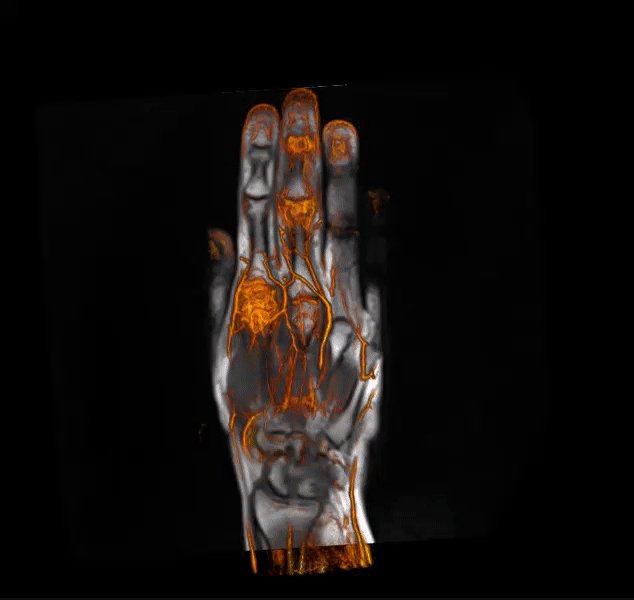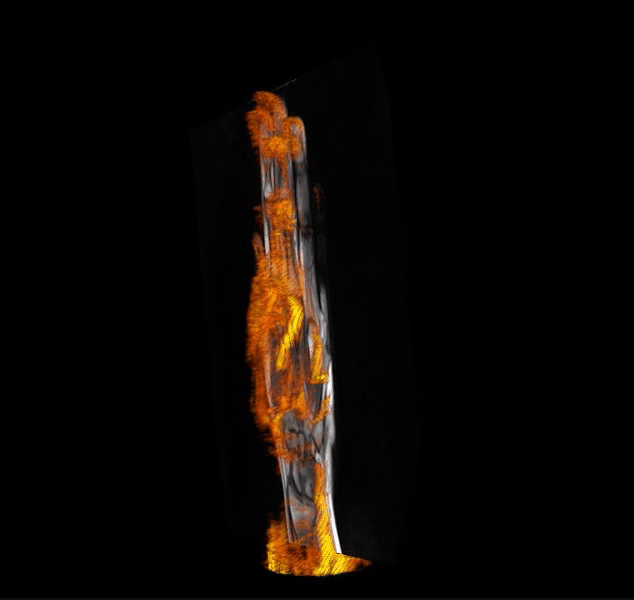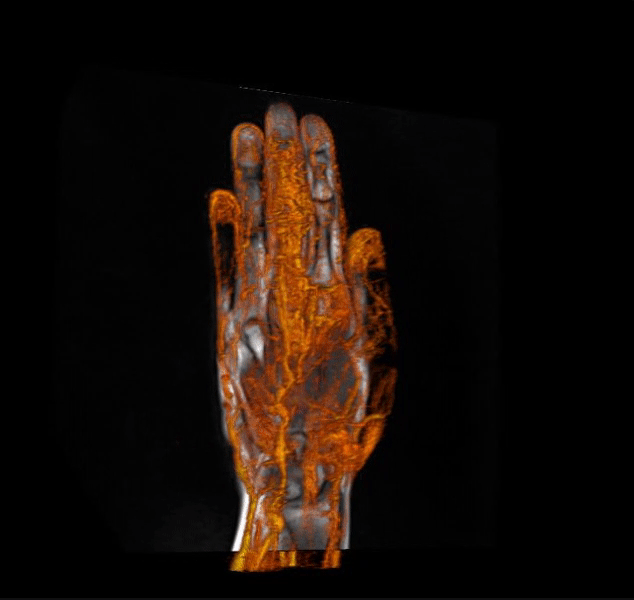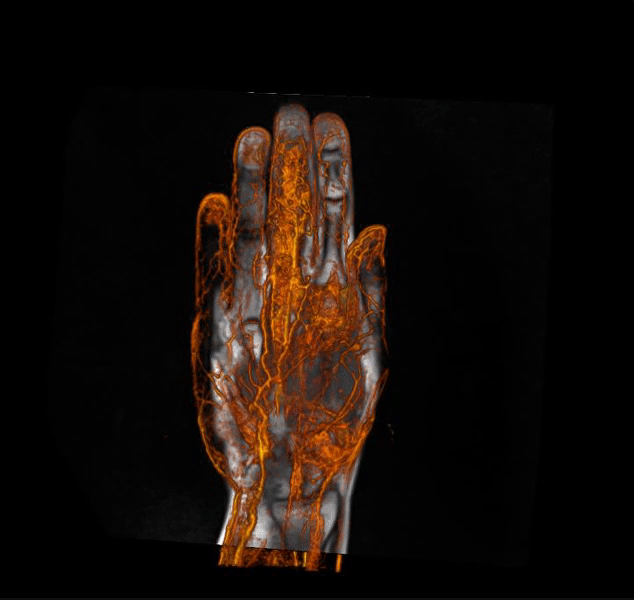
IAG helps clients in optimizing clinical trial designs with imaging-based endpoints that can accelerate go / no-go efficacy decisions for emerging therapeutics and enrich patient selection in musculoskeletal, oncology, immunology, rheumatology, neuroscience, CNS and rare disease trials.
IAG’s Medical and Scientific advisory teams will support trial design and protocol development.
Our Operational team and project managers are located in the UK, EU, USA, India and will support phase 1-3 trials and real-world studies with sites located globally.
We are here to create successful partnerships and to see more patients cured with the drugs, that we can help you to develop.